Tert-amylbenzene is a benzene derivative. At normal temperature and pressure, it is a colorless to pale yellow liquid with physical and chemical properties similar to those of toluene. It is insoluble in water but miscible with most organic solvents. Tert-amylbenzene can be prepared by the Friedel-Crafts alkylation reaction of benzene and tert-amyl alcohol. This substance has a relatively low boiling point and can be purified by distillation in industrial production. It is mainly used as a basic reagent in pharmaceutical chemistry. Some studies have reported that it can be used in the synthesis of the drug molecule amolprofen hydrochloride.
Production process
Method One
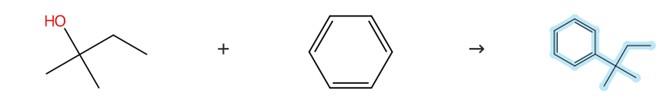
Figure 1 Synthesis method of tert-pentylbenzene
Add carborane silane cation [Me3Si(HCB11H5Br6)] (1.4 mg, 2.0 millimoles, 1.0 molar %) to benzene (0.5 ml), then add trimethylphenylsilane (66.1 mg, 0.44 millimoles, 2.2 equivalents) and 2, 2-dimethylpropyl 1-alcohol (17.6 mg) to the mixture. The mixture was stirred at 70°C for 24 hours (0.20 millimoles, 1.0 equivalent). After the reaction was completed, the reaction mixture was directly distilled and purified under vacuum to obtain the target product molecule tert-amylbenzene. [1]
Method Two
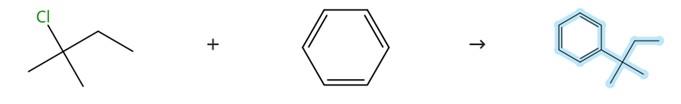
Figure 2 Synthesis method of tert-pentylbenzene
Some researchers have reported a production process of tert-pentylbenzene. The reported synthesis method uses amyl alcohol as the raw material to react with hydrochloric acid to generate tert-pentylchlorine, and then, under the action of a catalyst, reacts tert-pentylchlorine with toluene to produce tert-pentylbenzene products. The production process of tert-pentylbenzene using this method has the advantages of abundant raw materials, simple process operation and relatively high yield.
Purification method
Crude tert-pentylbenzene containing secondary pentylbenzene is dissolved in a non-polar solvent. After dissolution, a halogenating agent is added to carry out a halogenation reaction to obtain a mixed solution of halogenated secondary pentylbenzene and tert-pentylbenzene. (2) The mixed solution of halogenated secondary amylbenzene and tertiary amylbenzene obtained in step (1) was subjected to vacuum distillation to obtain the distilled concentrate. (3) The distillation concentrate obtained in step (2) of vacuum distillation is the purified tert-amylbenzene. The synthetic method reported in this paper provides a purification approach for high-purity tert-pentylbenzene, which is simple to operate and suitable for industrial production.










